Among the various approaches to ammonia synthesis, the lithium-mediated nitrogen reduction (Li-NRR) strategy is considered one of the most promising electrochemical methods, as it has the potential to overcome the high energy consumption and significant CO2 emissions associated with the traditional Haber–Bosch process. However, the currently achieved current densities remain insufficient to meet industrial requirements.
To address this challenge, Prof. Tao Cheng’s group at the Institute of Functional Nano & Soft Materials, Soochow University, in collaboration with Prof. Jun Li’s group at Shanghai Jiao Tong University, recently regulated the solid electrolyte interphase (SEI) structure and achieved an ammonia Faradaic efficiency as high as 98% at an industrial-level current density of 100 mA cm-2. In a continuous-flow electrochemical cell, the system operated stably for 40 hours, maintaining a Faradaic efficiency of 80%. High reaction rates and selectivity for nitrogen reduction were achieved under ambient conditions.
Theoretical calculations and experimental characterizations revealed that the SEI structure plays a critical role in the catalytic process. High-resolution transmission electron microscopy (HRTEM) and synchrotron X-ray spectroscopy confirmed a unique SEI architecture composed of an inner Li3N layer, a middle Li2CO3 layer, and an outer LiF layer. Theoretical results indicate that the LiF-rich outer layer enhances Li+ desolvation, while the Li2CO3-rich middle layer improves Li+ conductivity. This engineered SEI thus simultaneously enables efficient Li+ desolvation and migration. Kinetic modeling further demonstrates that this Li+ transport mechanism drives the highly efficient nitrogen reduction reaction.
Through this joint effort, the teams not only achieved efficient ammonia synthesis under ambient temperature and pressure, but also provided in-depth mechanistic insights into the critical role of the SEI, offering guidance for the rational design of catalytic systems, improved energy utilization, and progress toward carbon neutrality goals.
Dr. Qiang Zhang (postdoctoral fellow, Frontiers Science Center for Transformative Molecules, Shanghai Jiao Tong University), Dr. Huamin Li (visiting PhD student, Frontiers Science Center for Transformative Molecules), Peiping Yu (PhD student, Soochow University), and Pengyu Liu (PhD student, Frontiers Science Center for Transformative Molecules) are the co–first authors of the paper. Prof. Jun Li from Shanghai Jiao Tong University and Prof. Tao Cheng from Soochow University are the corresponding authors. This work was supported by multiple funding sources. Prof. Tao Cheng’s group received financial support from the Collaborative Innovation Center of Suzhou Nano Science and Technology, the National Natural Science Foundation of China (Grant No. 92472110 and 22173066), and the Natural Science Foundation of Jiangsu Province (BK20230065). The related work was published online in Science on February 12, 2026 (DOI: 10.1126/science.adw5462).
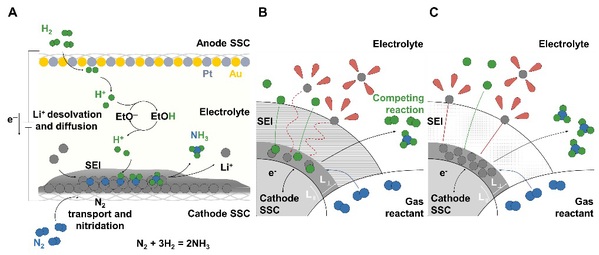
Fig. Limiting current in Li-mediated nitrogen electrolysis and ion-transport SEI film. (A) Schematic of N2 electrolysis in a continuous-flow electrolyzer. (B and C) Schematic illustrations of N2 electrolysis at the SEI with low and high ionic conductivity.
Link to paper:https://www.science.org/doi/10.1126/science.adw5462
Title:Enhanced Li-ion diffusion improves N2-to-NH3 current efficiency at 100 mA cm−2
Authors:Qiang Zhang#, Huamin Li#, Peiping Yu#, Pengyu Liu#, Ning Sun, Yiyan Wang, Chunlai Tu, Yiping Liu, Yan Wang, Xinyang Yue, Linlin Ma, Wen Wen, Jinyang Xu, Zhaofeng Liang, Jingyuan Ma, Fei Song, Zheng Liang, Hao Sun, Daishun Ling, Hongyan Liang, Feng Liu, Yongfeng Hu, Tao Cheng*, Jun Li*
Editor: Guo Jia